Late-Stage & Post-Market Services

Tailored manufacturing grades
Commercial GMP Manufacturing for Oligonucleotides
Streamlined Path to Market
We deliver cGMP manufacturing solutions for commercial oligonucleotide production, supporting your therapy through launch, post-launch supply, and lifecycle management. With 10 years of commercial experience and 9 commercial oligonucleotide products supported, BioSpring is the trusted global partner for scaling oligonucleotide-based therapeutics.

Where Quality Meets Scale
Global Commercial Supplier
- Partner of choice for the top 25 biotech & pharma companies
- 9 commercial oligonucleotide products supported
- FDA- and ANVISA- inspected
- Manufacturer of commercial oligonucleotide products marketed worldwide
 Proven Process Validation to Make You Market Ready
Proven Process Validation to Make You Market Ready
We offer proven process validation services to accelerate regulatory approval and derisk your product launch. Our dedicated teams for process validation and process characterization provide the expertise needed for late-stage development through commercialization. Our comprehensive, in-house validation master plan is designed to accelerate PPQ readiness. In addition, we support platform validation approaches to help reduce the validation effort and increase efficiency.
 Seamless Tech Transfer and Scalable Production
Seamless Tech Transfer and Scalable Production
Our experienced teams manage late-stage and commercial process and analytical method transfers with speed and precision. From process optimization and scale-up to comparability studies, we ensure your product is ready for commercial supply without disruption. With the ability to manufacture commercial batch sizes from grams to tens of kilograms, our facilities scale flexibly alongside your program’s growth.
 Global Supply with Proven Reliability
Global Supply with Proven Reliability
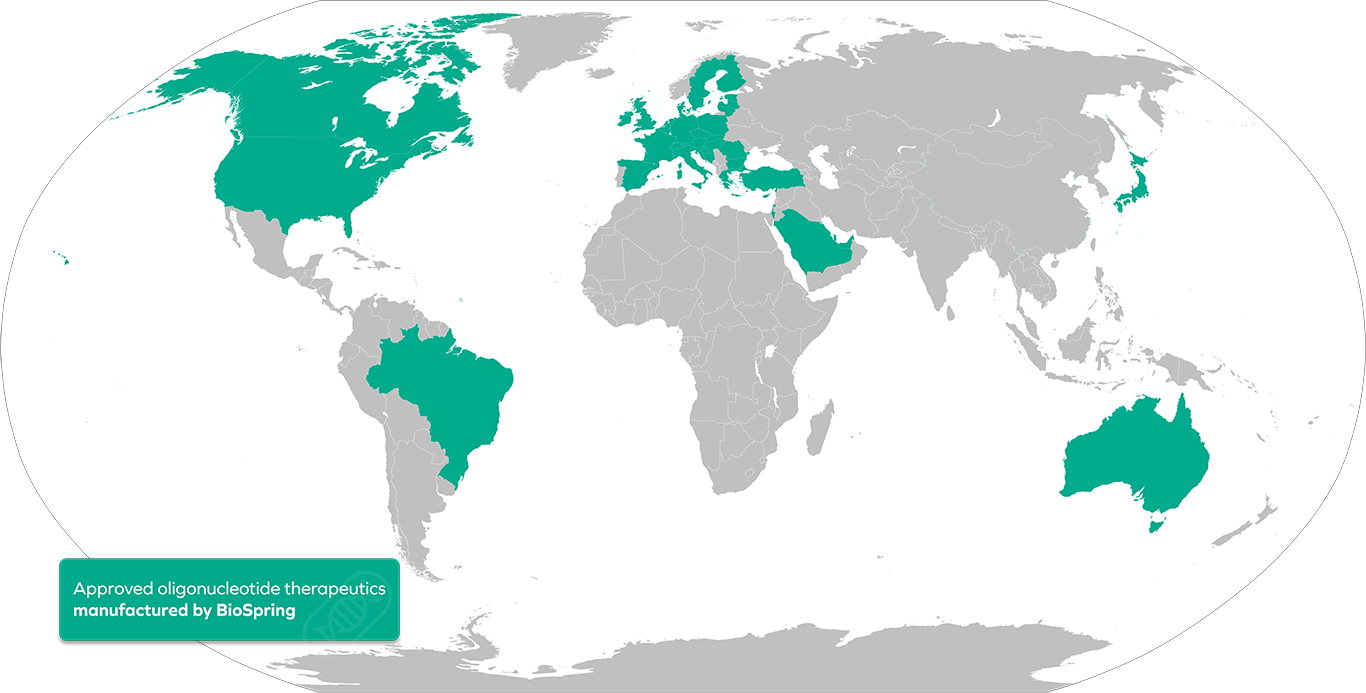
To date, BioSpring has successfully supported 9 commercial programs, marketed across the US, EU, UK, Japan, Brazil, Saudi Arabia, China, and more. Our global supply capabilities are built on a robust quality system and an exemplary regulatory track record, ensuring long-term reliability for marketed therapies.
 Regulatory Expertise to Accelerate Approval
Regulatory Expertise to Accelerate Approval
Our regulatory experts guide your team through the complexities of global submission requirements. We offer both the authoring of quality-related dossier sections, including Module 3, and the review of marketing applications such as NDAs, BLAs, MAAs, and J-NDAs. Additionally, we provide strategic input on risk assessments, Extractables & Leachables (E&L) assessments, and post-approval change management. Our services include strategies for implementation of ICH M7 requirements and preparation of statements. With decades of experience in oligonucleotide manufacturing and analysis, BioSpring is your partner for phase-appropriate, approval-ready execution.
Why BioSpring?
Ready for what's next?
How we start:
1. You share the basics: modality, sequence & target scale
2. Our team will evaluate project requirements & curate a project plan
3. You get a proposal & timeline: Scope, deliverables & costs
 Accelerate your success with us:
Accelerate your success with us:
Reduce CMC risk, keep timelines predictable, and progress confidently to submission and launch.
Learn more about our solutions